Lithium is a critical material for modern energy storage, yet extracting it efficiently from salt lake brines remains a major technological challenge. Conventional processes are time-consuming and suffer from significant lithium losses, while direct lithium extraction (DLE) technologies are often limited by the difficulty of separating lithium ions (Li+) from sodium ions (Na+), which share nearly identical physicochemical properties.
Membrane-based separation is widely considered a promising approach for DLE due to its continuous operation and scalability. However, conventional membranes rely mainly on size-based sieving, making it extremely challenging to distinguish Li+ from Na+. As a result, they often suffer from a long-standing trade-off between selectivity and permeance.
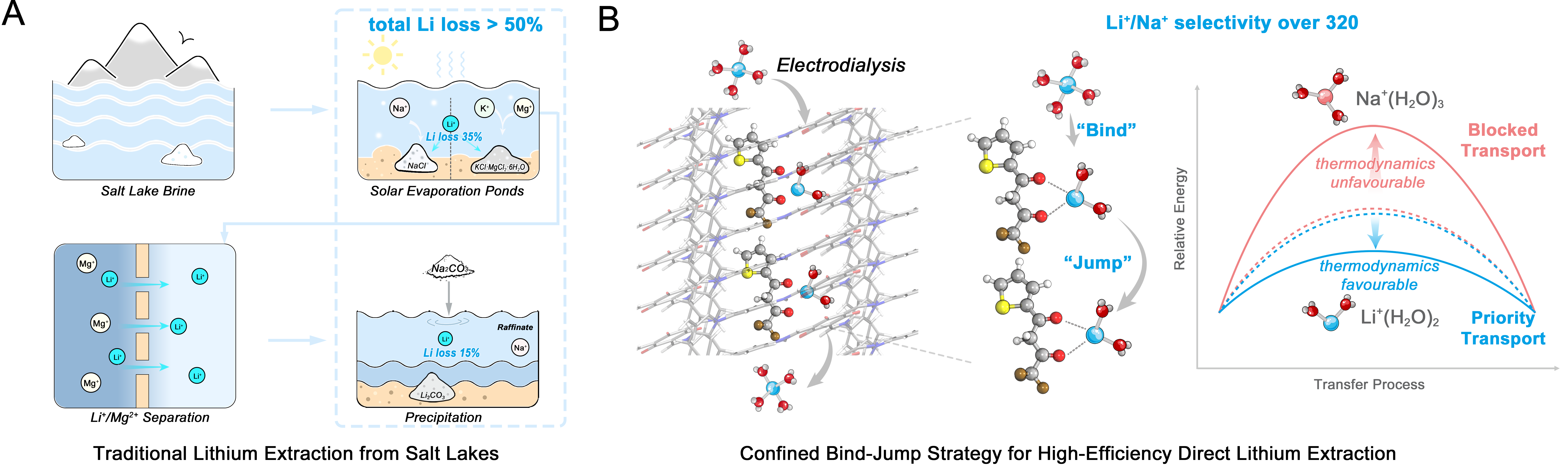
To address this bottleneck, a research team led by Prof. Zhong Liu from the Qinghai Institute of Salt Lakes (QISL), Chinese Academy of Sciences, has developed a new membrane-based strategy for highly efficient Li+/Na+ separation.
In a study published in Science Advances, the researchers report an “ion trap membrane” that enables both high lithium selectivity and fast ion transport—two properties that are typically difficult to achieve simultaneously.
The new design overcomes this limitation by introducing lithiophilic diketone molecules (HTTA) into the nanochannels of a covalent organic framework (COF) membrane. These molecules create a confined, hydrophobic environment that selectively interacts with Li+ ions.
Under an applied electric field, Li+ ions are preferentially captured within the channel and partially dehydrated. They then migrate via a directional hopping process between adjacent binding sites: a mechanism described by the team as “bind–jump” transport. In contrast, Na+ and K+ ions exhibit weaker interactions and higher migration resistance, leading to their effective exclusion.
As a result, the optimized membrane achieves a Li+ permeance of 143 mmol·m-2·h-1 and a Li+/Na+ selectivity of up to 320. Compared with unmodified COF membranes, this represents simultaneous improvements of approximately 2.5 times in permeance and over two orders of magnitude in selectivity, effectively overcoming the conventional trade-off limitation.
Importantly, the membrane also performs well in real salt lake brines from both China and abroad. A single-stage electrodialysis process reduces the Na+/Li+ ratio to about one-eighth of its initial value, demonstrating strong potential for practical deployment.
“This work shows that ion recognition within confined channels can fundamentally change how ions move through membranes,” said the researchers. “By coupling thermodynamic binding with kinetic transport, we provide a new pathway to break the traditional performance limits.”
The study offers a promising strategy for efficient and low-energy lithium recovery from complex brine systems, and may inspire the design of next-generation ion-selective membranes for a wide range of applications.
Bind-jump Strategy for high-efficiency direct lithium extraction (Image by QISL)
