Recently, the Solution Structure and Interface Research Group (led by Professor Zhou Yongquan) at the Qinghai Institute of Salt Lakes has made new progress in the selective separation of cesium resources in salt lakes. The research findings were published in Chem. Eng. J. (Top Tier 1) and J. Environ. Chem. Eng. (Top Tier 2), with the Qinghai Institute of Salt Lakes as the first affiliation and Assistant Professor Ikram Muhammad as the first author.
The rare alkali metal cesium (Cs) is an important strategic resource with considerable reserves in salt lake brines. Research into processes for the extraction and separation of Cs from such brines holds significant application potential. However, no method currently exists for the large-scale extraction of Cs from salt lake brines. Owing to the highly similar physicochemical properties of alkali metal ions, Cs⁺ often coexists with other ions, such as K⁺, Na⁺, Ca²⁺, and Mg²⁺, making the efficient extraction and separation of Cs⁺ from salt lake brines challenging. Ion exchange and adsorption are considered ideal strategies for the separation of this dispersed element due to their cost-effectiveness, operational simplicity, and low secondary waste generation. Nevertheless, traditional methods face limitations, including insufficient selectivity and poor structural stability in aqueous environments, which restrict their widespread application.
Over the past two decades, metal–organic framework (MOF) derived materials have emerged as highly promising candidates for adsorption applications due to their controllable morphology and tunable porosity. However, conventional high-temperature calcination methods (400–900 °C), typically used to convert MOFs into metal oxides or sulfides, often compromise structural integrity, leading to particle aggregation, reduced specific surface area, and embedded active sites, that ultimately lower the materials adsorption capacity. To overcome these limitations, the Solution Structure and Interface Research Group (led by Professor Zhou Yongquan) at the Qinghai Institute of Salt Lakes, Chinese Academy of Sciences, proposed a novel solvothermal etching strategy. This innovative approach enables precise control over the crystallinity of MOF-derived materials by optimizing heat treatment conditions. It enhances porosity and facilitates ion diffusion while also allowing targeted engineering of material properties through strategies such as constructing defect structures, regulating coordination sites, increasing specific surface area, and incorporating heteroatom dopants (e.g., nitrogen doping). These synergistic modifications effectively modulate the material's electronic conductivity and substantially enhance its ion capture capacity.
Based on this strategy, the research team converted dodecahedral ZIF-8 MOF into carbon-free, nitrogen-doped zinc oxide rods (HNZOR) and subsequently transformed them into zinc sulfide spheres (HNZSS). Compared with traditional calcination methods, this approach effectively suppressed metal agglomeration, fully exposed active sites, and generated amorphous regions with high surface area. These characteristics synergistically enhanced mass transfer capability and created strong binding interactions within the adsorbent framework, resulting in excellent adsorption performance. Moreover, the K⁺-activated adsorbents (KNZOR and KZNSS) exhibited fast adsorption kinetics and high adsorption capacities (275 mg/g and 302 mg/g, respectively), overcoming interference from competing ions and demonstrating exceptional selectivity for Cs⁺ in complex environments. In real water samples, they showed outstanding adsorption efficiency, with distribution coefficients (Kd) reaching 2061.85 mL/g for KNZOR and 2578.31 mL/g for KZNSS. In practical column operations, KNZOR and KZNSS achieved near-quantitative (approximately 99%) recovery of Cs⁺ under continuous flow conditions. The research findings were published in Chem. Eng. J. (Top Tier 1) under the title "In-situ sequential topotactic transformation of ZIF-8 to N-doped Zn-oxide/sulfide with defect engineering for efficient radiocesium capture."
This study reveals a new pathway for the preparation of inorganic metal oxides/sulfides from metal–organic framework precursors and achieves selective capture of cesium ions from salt lakes. The elucidated recognition and adsorption mechanisms, together with the expanded MOF material library, provide guidance for the development of next-generation inorganic adsorbent materials for the efficient separation of target radionuclides, and are of great significance for the green development and utilization of dispersed element resources in salt lakes.
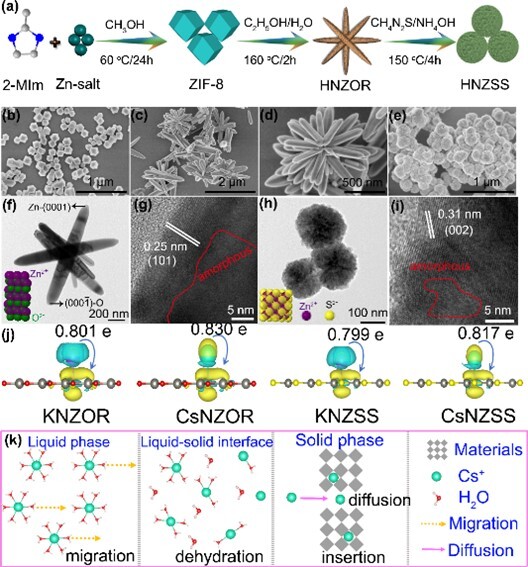
Figure 1. Material preparation; SEM, TEM, and HRTEM images; DFT calculations; and a schematic diagram of the cesium adsorption process.
In a separate study, a robust K⁺-directed metal sulfide (KCoZnS) nanosheet (NS) with high porosity and large specific surface area was derived from a bimetallic metal–organic framework (MOF) via layer-by-layer self-assembly, enabling rapid and selective capture of Cs⁺ ions in acidic media, with excellent stability and radiation resistance. KCoZnS exhibited outstanding ion exchange capacity (qe ≈ 400 mg/g), high removal efficiency (approximately 93%), and a large distribution coefficient (3.09 × 10⁴ mL/g). Moreover, the ion exchange process was facile and reversible, achieving an elution efficiency exceeding 99%. The interaction between Cs⁺ and the interlayer region of KCoZnS was confirmed and elucidated through XRD, XPS, EDS, and density functional theory (DFT) calculations. The high adsorption capacity and exceptional selectivity of KCoZnS for Cs⁺ can be attributed to its superior ion exchange performance, structural flexibility, and the coordination between the soft base S²⁻ and the soft acid Cs⁺ (i.e., Cs–S bonds). This study demonstrates a new approach for efficient Cs⁺ capture using MOF-derived metal sulfides. The research findings were published in J. Environ. Chem. Eng. (Top Tier 2) under the title "Metal organic frameworks derived novel metal sulfide (KCoZnS) nanosheet as an efficient collector for cesium ions."
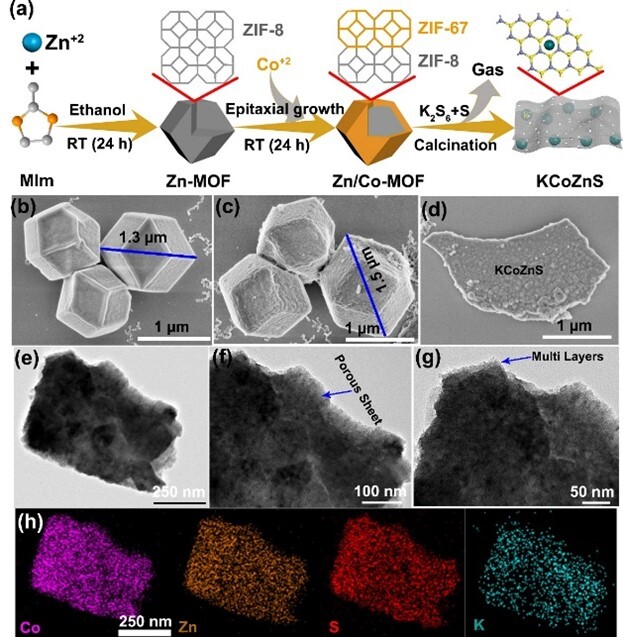
Figure 2. Synthesis scheme and morphology of ZIF-8, MLF, and KCoZnS.
This work was supported by the Outstanding Youth Foundation Project of Qinghai Province (Grant No. 2025-ZJ-967J), the Qinghai Institute of Salt Lakes, Chinese Academy of Sciences (Grant No. E355HX01), and the Youth Team Project of the Qinghai Institute of Salt Lakes, Chinese Academy of Sciences (Grant No. ISLJCTD-2022-04)
Article links:
https://doi.org/10.1016/j.cej.2026.172943
https://doi.org/10.1016/j.jece.2025.119802
